RELEASE
ARTICLE
The FDA has approved Vybrique (IBSA), an oral film formulation of the phosphodiesterase type 5 (PDE5) inhibitor sildenafil, for treatment of erectile dysfunction (ED). Sildenafil oral tablets (Viagra, and generics) have been available since 1998. The manufacturer is promoting the new product as a "discreet, on-the-go" option that can be taken without water.

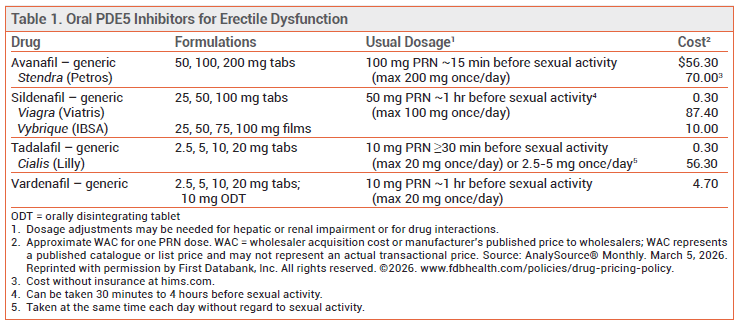
STANDARD TREATMENT — Oral PDE5 inhibitors (see Table 1) are generally used for initial treatment of ED. An FDA-approved, nonmedicated alcoholbased topical gel (Eroxon), which is available over the counter, improved erection response in about 60% of men who used it in clinical trials.1 Other treatment options include vacuum-assisted erection devices, intraurethral or intracavernosal alprostadil, and penile implants.2 Low-intensity extracorporeal shockwave therapy (not approved by the FDA) has gained popularity for treatment of ED, but quality evidence demonstrating its long-term effectiveness is lacking.3
MECHANISM OF ACTION — Sildenafil is a selective inhibitor of PDE5. Inhibition of PDE5 increases cyclic guanosine monophosphate (cGMP) levels in the corpus cavernosum, which leads to smooth muscle relaxation and inflow of blood.
CLINICAL STUDIES — FDA approval of sildenafil oral film was based on earlier trials with sildenafil tablets and one placebo-controlled trial (summarized in the package insert) in 475 men with ED in which the rate of successful attempts at sexual intercourse over 12 weeks was significantly higher in men who took sildenafil oral film.
ADVERSE EFFECTS — Adverse effects of sildenafil include headache, flushing, dyspepsia, sudden vision loss, nasal congestion, and back pain. Prolonged erection/priapism can occur. Nonarteritic anterior ischemic optic neuropathy (NAION), retinal detachment, sudden hearing loss, decreased blood pressure, cardiovascular or cerebrovascular events, and syncope have been reported with PDE5 inhibitors.
DRUG INTERACTIONS — Like the other PDE5 inhibitors, sildenafil is metabolized by CYP3A4. Inhibitors of CYP3A4 can markedly increase sildenafil serum concentrations.4 All PDE5 inhibitors can increase the hypotensive effects of organic nitrates such as nitroglycerin and guanylate cyclase stimulators such as riociguat; they are contraindicated for use in patients taking these drugs. They may also potentiate the antihypertensive effects of alpha blockers, other antihypertensive drugs, and alcohol.
DOSAGE AND ADMINISTRATION — Vybrique will be available for home delivery. It is supplied in cartons containing 4 or 8 individually packaged oral films, each containing 25, 50, 75, or 100 mg of sildenafil. The film should be placed on the tongue and swallowed with saliva once it has dissolved; it should not be cut or chewed. The recommended dosage is 50 mg taken as needed about one hour (30 minutes to 4 hours) before sexual activity; the dose can be increased to a maximum of 100 mg. Only one dose should be taken each day. The recommended starting dosage is 25 mg in patients who are taking a strong CYP3A4 inhibitor or alpha blocker, have hepatic or severe renal impairment, or are >65 years old.
- Eroxon – an OTC gel for erectile dysfunction. Med Lett Drugs Ther 2024; 66:181.
- AL Burnett et al. Erectile dysfunction: AUA guideline. J Urol 2018; 200:633. doi:10.1016/j.juro.2018.05.004
- Shockwave therapy for erectile dysfunction. Med Lett Drugs Ther 2023; 65:190.
- Inhibitors and inducers of CYP enzymes, P-glycoprotein, and other transporters. Med Lett Drugs Ther 2023 January 25 (epub). Available at: www.medicalletter.org/downloads/CYP_PGP_Tables.pdf.