ISSUE1726
- Mark Abramowicz, M.D., President has disclosed no relevant financial relationships.
- Jean-Marie Pflomm, Pharm.D., Editor in Chief has disclosed no relevant financial relationships.
- Michael Viscusi, Pharm.D., Associate Editor has disclosed no relevant financial relationships.
- Review the efficacy and safety of Penmenvy, a pentavalent meningococcal vaccine.
- Description: The second pentavalent meningococcal vaccine (Penbraya was the first).
- Indication: FDA-licensed for prevention of invasive meningococcal disease caused by Neisseria meningitidis serogroups A, B, C, W, or Y in persons 10-25 years old.
- Efficacy: Compared to separate administration of MenACWY and MenB vaccines, seroresponse rates with Penmenvy were similar against MenACWY and similar or slightly inferior against MenB.
- Adverse Effects: Injection-site reactions, fatigue, headache, nausea, myalgia, and arthralgia can occur.
- Dosage: Two 0.5-mL IM doses given 6 months apart.
- Conclusion: Pentavalent vaccination is recommended only for persons ≥10 years old who would otherwise have received both a MenACWY and a MenB vaccine at the same visit. How Penmenvy compares to Penbraya is unknown.
Outline
Table |
The FDA has licensed the pentavalent vaccine Penmenvy (GSK) for prevention of invasive meningococcal disease caused by Neisseria meningitidis serogroups A, B, C, W, or Y (MenABCWY) in persons 10-25 years old. Penmenvy is the second pentavalent meningococcal vaccine to become available in the US; Penbraya was licensed in 2023.1

MENINGOCOCCAL DISEASE — Six serogroups of N. meningitidis (A, B, C, W, X, and Y) cause most cases of invasive meningococcal disease worldwide. Most US cases are caused by serogroups B, C, and Y. Infection rates are highest in infants, adolescents/young adults, and adults >80 years old.2
THE NEW VACCINE — Penmenvy contains the antigenic components of Menveo, a polysaccharide conjugate quadrivalent MenACWY vaccine, and Bexsero, a recombinant protein-based MenB vaccine.
CLINICAL STUDIES — FDA licensure of Penmenvy was based on the results of two randomized, observer-blind immunogenicity trials (Trials 1 and 2).3,4
Trial 1 included 3651 persons 10-25 years old who had never received a MenB vaccine and had either never received a MenACWY vaccine (87%) or received a single MenACWY vaccine dose >4 years earlier (13%). Patients were given a 2-dose series of Penmenvy (0 and 6 months), a 2- or 3-dose series of Bexsero (either 0 and 6 or 0, 2, and 6 months), or a single dose of Menveo.
Seroresponse rates against MenACWY 1 month after the final dose were noninferior with Penmenvy compared to Menveo (A: 96.8% vs 85.6%; C: 97.2% vs 50.0%; W: 97.0% vs 61.7%; Y: 96.7% vs 69.7%). Seroresponse rates with Penmenvy were noninferior to those with two doses of Bexsero against the MenB antigens NadA (92.7% vs 95.9%) and fHbp (73.2% vs 78.1%), but not against NHBA (61.8% vs 69.7%) and PorA P1.4 (42.2% vs 58.3%).3
In Trial 2, 1250 persons 15-25 years old who had received one dose of a MenACWY vaccine ≥4 years earlier were given a 2-dose series of Penmenvy (0 and 6 months) or a single dose of Menveo. Seroresponse rates against MenACWY 1 month after the final dose were noninferior with Penmenvy compared to Menveo (A: 95.8% vs 95.2%; C: 94.5% vs 94.0%; W: 95.6% vs 93.9%; Y: 95.0% vs 94.4%).4
ADVERSE EFFECTS — Adverse effects of Penmenvy in clinical trials were similar to those with Bexsero and included fatigue, headache, nausea, myalgia, arthralgia, and injection-site pain, erythema, swelling, and induration. Syncope, dizziness, lymphadenopathy, and hypersensitivity reactions have been reported. Administration of conjugate MenACWY vaccines has been associated with Guillain-Barré syndrome.
PREGNANCY AND LACTATION — No data are available on the use of Penmenvy in pregnant or lactating women. MenACWY vaccination during pregnancy appears to be safe, but data on MenB vaccination during pregnancy are limited. Following licensure of Penbraya, the CDC stated that MenB vaccination should be deferred until after pregnancy unless risk factors for infection are present and the benefits are determined to outweigh the risks. Pregnant women requiring MenACWY vaccination should generally receive a quadrivalent vaccine.5 Breastfeeding women can receive any meningococcal vaccine.6
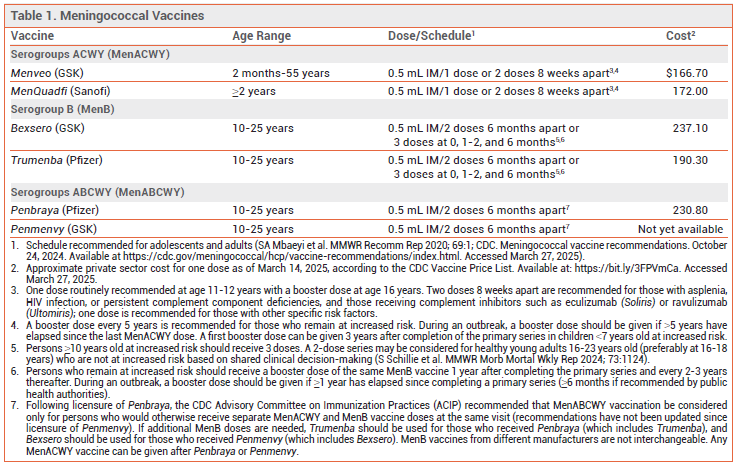
DOSAGE AND ADMINISTRATION — Penmenvy consists of a vial containing the MenACWY component (lyophilized powder) and a prefilled syringe containing the MenB component (suspension). The powder must be reconstituted with the suspension to form a single 0.5-mL dose. Components should be stored in a refrigerator, and the vaccine should be administered immediately after reconstitution. The recommended dosage is two 0.5-mL IM doses given 6 months apart.
ACIP RECOMMENDATIONS — The CDC’s Advisory Committee on Immunization Practices (ACIP) updated its recommendations for use of MenACWY and MenB vaccines in 2020.7
MenACWY (Menveo, MenQuadfi) – Routine vaccination with MenACWY is recommended for adolescents 11-18 years old, with a first dose given at 11-12 years and a booster dose given at 16 years. Two doses given at least 8 weeks apart and a booster dose every 5 years are recommended for adults with HIV infection, functional or anatomic asplenia, or persistent complement component deficiencies (including impairment induced by complement inhibitors). One dose of vaccine is recommended for adults who travel to or live in countries where meningococcal disease is hyperendemic or epidemic, persons who are at risk from a meningococcal disease outbreak attributed to serogroups A, C, W, or Y, microbiologists routinely exposed to N. meningitidis, military recruits, and first-year college students living in dormitories (if they did not receive a dose at age ≥16 years); revaccination every 5 years is recommended for those who remain at risk.
MenB (Bexsero, Trumenba) – MenB vaccine is recommended for adults with functional or anatomic asplenia, persons with persistent complement component deficiencies (including impairment induced by complement inhibitors), those who are at risk from a meningococcal outbreak attributed to serogroup B, and microbiologists routinely exposed to N. meningitidis. It may be considered for persons 16 through 23 years old (preferably age 16-18 years) who do not have risk factors for meningococcal disease, but may be at increased risk by attending college (e.g., college students living in dormitories). Persons who remain at increased risk should receive a booster dose 1 year after completing the primary series and every 2-3 years thereafter. During an outbreak, previously vaccinated persons should receive a single booster dose if ≥1 year has elapsed since completion of the primary series (≥6 months if recommended by public health authorities). MenB vaccines from different manufacturers are not interchangeable; vaccines from the same manufacturer must be used for all doses.8
MenABCWY (Penbraya, Penmenvy) – Following approval of Penbraya in 2023, the ACIP recommended that MenABCWY vaccination be considered only for persons ≥10 years old who would otherwise receive separate MenACWY and MenB vaccine doses at the same visit.5 The ACIP is scheduled to discuss recommendations for Penmenvy in April 2025.9
CONCLUSION — Penmenvy is the second pentavalent meningococcal vaccine containing Neisseria meningitidis serogroups A, B, C, W, and Y (MenABCWY) to be licensed by the FDA. How it compares to Penbraya in efficacy and safety remains to be determined. Use of Penmenvy or Penbraya reduces the total number of injections required for full vaccination against these five serogroups, which may improve compliance. Pentavalent vaccines are recommended only for persons ≥10 years old who would otherwise receive both a MenACWY and a MenB vaccine at the same visit.
- Penbraya: a pentavalent meningococcal vaccine. Med Lett Drugs Ther 2024; 66:43.
- CDC. Meningococcal disease surveillance and trends. November 12, 2024. Available at: https://bit.ly/4kN7eVC. Accessed March 27, 2025.
- T Nolan et al. Breadth of immune response, immunogenicity, reactogenicity, and safety for a pentavalent meningococcal ABCWY vaccine in healthy adolescents and young adults: results from a phase 3, randomised, controlled observer-blinded trial. Lancet Infect Dis 2024 December 5 (epub). doi:10.1016/s1473-3099(24)00667-4
- T Nolan et al. Immunogenicity, reactogenicity, and safety of a pentavalent meningococcal ABCWY vaccine in adolescents and young adults who had previously received a meningococcal ACWY vaccine: a phase 3, randomized controlled clinical study. Clin Infect Dis 2024 December 26 (epub). doi:10.1093/cid/ciae622
- JP Collins et al. Use of the Pfizer pentavalent meningococcal vaccine among persons aged ≥10 years: recommendations of the advisory committee on immunization practices ― United States, 2023. MMWR Morb Mortal Wkly Rep 2024; 73:345. doi:10.15585/mmwr.mm7315a4
- Drugs and Lactation Database (LactMed) [Internet]. Bethesda (MD): National Institute of Child Health and Human Development; 2006-. Meningococcal vaccines. [Updated 2024 Aug 15]. Available at: https://bit.ly/41Z4kWq. Accessed March 27, 2025.
- SA Mbaeyi et al. Meningococcal vaccination: recommendations of the Advisory Committee on Immunization Practices, United States, 2020. MMWR Recomm Rep 2020; 69:1. doi:10.15585/mmwr.rr6909a1
- S Schillie et al. New dosing interval and schedule for the Bexsero MenB-4C vaccine: updated recommendations of the Advisory Committee on Immunization Practices — United States, October 2024. MMWR Morb Mortal Wkly Rep 2024; 73:1124. doi:10.15585/mmwr.mm7349a3
- CDC Advisory Committee on Immunization Practices. ACIP meeting information. March 24, 2025. Available at: https://bit.ly/3R2ICdN. Accessed March 27, 2025.