RELEASE
ARTICLE
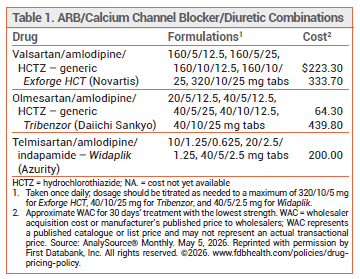
The FDA has approved Widaplik (Azurity), a fixed-dose combination of the angiotensin receptor blocker (ARB) telmisartan, the dihydropyridine calcium channel blocker amlodipine, and the thiazide-like diuretic indapamide, for treatment of hypertension in adults. Telmisartan, amlodipine, and indapamide are also available alone and in various combinations for treatment of hypertension. Two other three-drug combinations containing an ARB, amlodipine, and hydrochlorothiazide (Exforge HCT and Tribenzor) have been available for years for treatment of hypertension (see Table 1).1

STANDARD TREATMENT ― A thiazide or thiazide-like diuretic, an angiotensin-converting enzyme (ACE) inhibitor or ARB, and a long-acting dihydropyridine calcium channel blocker, alone or in two-drug combinations, is generally recommended for initial treatment of hypertension. Most patients require multiple drugs from different classes for blood pressure control.2
CLINICAL STUDIES ― FDA approval of Widaplik was based on the results of two double-blind trials in patients with hypertension. In the first trial, 295 patients taking ≤1 antihypertensive drug were randomized to receive telmisartan/amlodipine/indapamide (10/1.25/0.625 mg or 20/2.5/1.25 mg) or placebo after a 2-week washout period. The placebo-corrected mean change from baseline in home systolic blood pressure (SBP) at week 4, the primary endpoint, was -7.3 mm Hg with the lower dose and -10.4 mm Hg with the higher dose; the differences between the active drug and placebo were statistically significant.3
In the second trial, 1385 patients taking up to 3 antihypertensive drugs were switched to telmisartan 20 mg/amlodipine 2.5 mg/indapamide 1.25 mg during a 4-week run-in period. Patients were then randomized to continue the three-drug regimen or switch to a two-drug regimen (telmisartan 20 mg/amlodipine 2.5 mg, telmisartan 20 mg/indapamide 1.25 mg, or amlodipine 2.5 mg/indapamide 1.25 mg). At week 6, the doses of each component in all groups were doubled. The mean change from baseline in home SBP at week 12 was significantly greater with the three-drug combination than with the two-drug combinations (-4.0 mm Hg vs +1.4 mm Hg with telmisartan/amlodipine, -1.5 mm Hg with telmisartan/indapamide, and +0.5 mm Hg with amlodipine/indapamide).4
In a double-blind trial, 1670 patients with a history of intracerebral hemorrhage and a systolic blood pressure of 130 to 160 mm Hg were treated with a once-daily pill containing telmisartan 20 mg, amlodipine 2.5 mg, and indapamide 1.25 mg in addition to standard care (including other antihypertensive drugs). After 2 weeks, they were randomized to continue the three-drug combination or switch to placebo. At a median follow-up of 2.5 years, the rate of first recurrent stroke, the primary endpoint, was significantly lower in those taking the three-drug combination (4.6% vs 7.4%). The mean systolic blood pressure during follow-up (127 mm Hg vs 138 mm Hg) and the rate of major cardiovascular events (6.6% vs 9.8%) were also lower with the three-drug combination.5
ADVERSE EFFECTS ― The most common adverse effect of Widaplik in clinical trials was symptomatic hypotension. Hypokalemia, hyponatremia, hypomagnesemia, and hyperuricemia can occur. Serum electrolytes and glucose concentrations should be monitored periodically during treatment. Sulfonamide derivatives such as indapamide can cause acute angle-closure glaucoma and elevations in intraocular pressure; Widaplik should be discontinued in patients who experience decreased visual acuity or ocular pain.
Widaplik is contraindicated for use in patients with known hypersensitivity to sulfonamide derivatives and in those with diabetes who are also taking the direct renin inhibitor aliskiren (Tekturna, and others).
DOSAGE AND ADMINISTRATION ― The recommended starting dosage of Widaplik is 10/1.25/0.625 mg or 20/2.5/1.25 mg once daily. The dosage can be increased after 2 weeks to a maximum of 40/5/2.5 mg once daily. Some expert clinicians use maximum doses of each individual component (80/10/5 mg) for treatment-resistant hypertension. The tablets should be swallowed whole; they should not be crushed, chewed, or split.
CONCLUSION ― In clinical trials, Widaplik, a three-drug combination of the angiotensin receptor blocker (ARB) telmisartan, the dihydropyridine calcium channel blocker amlodipine, and the thiazide-like diuretic indapamide, was more effective than combinations containing two of its components at the same dosages. Widaplik offers a convenient alternative to taking the three components separately. Whether it offers any advantage over Exforge HCT or Tribenzor, which contain an ARB, a calcium channel blocker, and hydrochlorothiazide and are available generically, remains to be established.
- Drugs for hypertension. Med Lett Drugs Ther 2024; 66:81.
- DW Jones et al. 2025 AHA/ACC/AANP/AAPA/ABC/ACCP/ACPM/AGS/AMA/ASPC/NMA/PCNA/SGIM guideline for the prevention, detection, evaluation and management of high blood pressure in adults: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Hypertension 2025; 82:e212. doi:10.1161/hyp.0000000000000249
- A Rodgers et al. Efficacy and safety of a novel low-dose triple single-pill combination compared with placebo for initial treatment of hypertension. J Am Coll Cardiol 2024; 84:2393. doi:10.1016/j.jacc.2024.08.025
- A Rodgers et al. Efficacy and safety of a novel low-dose triple single-pill combination of telmisartan, amlodipine and indapamide, compared with dual combinations for treatment of hypertension: a randomised, double-blind, active-controlled, international clinical trial. Lancet 2024; 404:1536. doi:10.1016/s0140-6736(24)01744-6
- The Trident Research Group. Three low-dose antihypertensive agents in a single pill after intracerebral hemorrhage. N Engl J Med 2026; 394:1571. doi:10.1056/nejmoa2515043