ISSUE1621
- Mark Abramowicz, M.D., President: no disclosure or potential conflict of interest to report
- Jean-Marie Pflomm, Pharm.D., Editor in Chief: no disclosure or potential conflict of interest to report
- Brinda M. Shah, Pharm.D., Consulting Editor: no disclosure or potential conflict of interest to report
- Michael Viscusi, Pharm.D., Associate Editor: no disclosure or potential conflict of interest to report
- Review the efficacy and safety of bamlanivimab and etesevimab for COVID-19.
Revised 9/23/2021: As of September 2, 2021, distribution of bamlanivimab and etesevimab has resumed in all US jurisdictions (https://bit.ly/3AynAdr).
Revised 7/1/2021: On June 25, 2021, federal distribution of bamlanivimab and etesevimab was suspended because an increasing percentage of COVID-19 cases in the US are being caused by SARS-CoV-2 variants that are resistant to the drugs (https://bit.ly/3dsDH2I).
- Mechanism of Action
- Clinical Studies
- Variants
- Adverse Effects
- Dosage and Administration
- Availability
- Conclusion
- References
Table
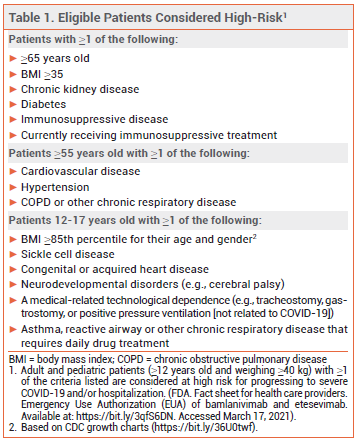
The FDA has issued an Emergency Use Authorization (EUA) for Lilly's investigational monoclonal antibodies bamlanivimab (LY-CoV555) and etesevimab (LYCoV016) to be administered together for treatment of mild to moderate COVID-19 in patients ≥12 years old who weigh ≥40 kg and are at high risk of progressing to severe disease and/or hospitalization (see Table 1).1 Bamlanivimab received an EUA for use as monotherapy in such patients in November 2020.2 Regeneron's investigational monoclonal antibodies casirivimab (REGN10933) and imdevimab (REGN10987) are also authorized for use together for the same indication.3

Use of monoclonal antibodies has been associated with worse clinical outcomes in hospitalized patients with COVID-19 who require high-flow oxygen or mechanical ventilation. Bamlanivimab and etesevimab are not authorized for use in patients who are hospitalized for COVID-19 or require oxygen therapy because of COVID-19.
MECHANISM OF ACTION — Bamlanivimab and etesevimab bind to different but overlapping sites on the receptor binding domain of the spike protein of SARS-CoV-2, blocking its attachment to the human ACE2 receptor.
CLINICAL STUDIES — Issuance of the EUA for bamlanivimab and etesevimab was based on the results of two randomized, double-blind trials in outpatients with mild to moderate COVID-19.
The first trial (BLAZE-1) had a phase 2 and a phase 3 component. In the phase 2 component, 612 adult patients (42% high risk) received a single infusion of bamlanivimab 2800 mg plus etesevimab 2800 mg, bamlanivimab alone (700 mg, 2800 mg, or 7000 mg), or placebo. The mean decrease from baseline in log SARS-CoV-2 viral load at day 11, the primary endpoint, was significantly greater with bamlanivimab plus etesevimab than with placebo (-4.37 vs -3.80). None of the doses of bamlanivimab alone significantly reduced viral load at day 11 compared to placebo.4
In the phase 3 component of BLAZE-1 (unpublished; summarized in the FDA Fact Sheet), 1035 high-risk adults received either bamlanivimab 2800 mg plus etesevimab 2800 mg or placebo. The primary endpoint, hospitalization due to COVID-19 or death from any cause by day 29, occurred significantly less often with the antibodies than with placebo (2.1% vs 6.6%; HR 0.32; NNT 22.5). No deaths occurred in the antibody group, compared to 10 in the placebo group (p <0.001).5,6
In the second trial (BLAZE-4; unpublished; summarized in the FDA Fact Sheet), 259 patients 18-65 years old with a BMI <35 kg/m2 received either bamlanivimab 2800 mg plus etesevimab 2800 mg or bamlanivimab 700 mg plus etesevimab 1400 mg. The mean change in SARS-CoV-2 viral load was similar with the two doses of the combination. The FDA selected 700 mg/1400 mg as the authorized dose for use of bamlanivimab and etesevimab together, which prompted the manufacturer to study this dosage in a new cohort of BLAZE-1.5
In the new BLAZE-1 cohort (unpublished; summarized in a press release), 769 high-risk patients ≥12 years old received either bamlanivimab 700 mg plus etesevimab 1400 mg or placebo. The rate of hospitalization due to COVID-19 or death from any cause by day 29 was significantly lower with the antibodies than with placebo (0.8% vs 5.4%; HR 0.13; NNT 21.5). No deaths occurred in the antibody group, compared to 4 in the placebo group.6
No studies directly comparing bamlanivimab and etesevimab with casirivimab and imdevimab are available.
VARIANTS — In BLAZE-1, treatment-emergent SARS-CoV-2 variants developed less frequently in patients who received bamlanivimab plus etesevimab than in those who received bamlanivimab alone. The combination is not active against the Omicron variant of SARS-CoV-2. It retains activity against the Delta variant of the virus.5
ADVERSE EFFECTS — Hypersensitivity reactions including anaphylaxis have been reported with use of bamlanivimab with and without etesevimab.
DOSAGE AND ADMINISTRATION — Bamlanivimab and etesevimab are supplied separately in 700 mg/20 mL vials, but when used together, they must be administered as a single IV infusion; the authorized dose is 700 mg (one vial) of bamlanivimab and 1400 mg (two vials) of etesevimab. The antibodies should be diluted in 50, 100, 150, or 250 mL of normal saline and infused at a maximum rate of 310 mL/hr (266 mL/hr when mixed with 250 mL normal saline for patients weighing <50 kg) immediately after preparation. If immediate use is not possible, the solution can be refrigerated for up to 24 hours or left at room temperature for up to 7 hours, including infusion time. If refrigerated, the solution should be allowed to sit at room temperature for 20 minutes before use.
Bamlanivimab and etesevimab should be administered as soon as possible after a positive SARS-CoV-2 test result and within 10 days of COVID-19 symptom onset. Patients should be treated in a facility staffed and equipped to manage anaphylaxis and they should be monitored for hypersensitivity reactions during administration of the drug and for at least 1 hour after completion of the infusion.
AVAILABILITY — Bamlanivimab and etesevimab will be allocated to state health departments by the US Department of Health and Human Services (HHS) based on case counts and severity of outbreaks. State health departments will then be responsible for allocating the antibodies to local health facilities. The manufacturer plans to produce 1 million doses of etesevimab for use with bamlanivimab by mid-2021.7
CONCLUSION — The FDA has issued an Emergency Use Authorization (EUA) for the monoclonal antibodies bamlanivimab (LY-CoV555) and etesevimab (LY-CoV016) to be administered together by IV infusion for treatment of mild to moderate COVID-19. Administration of the antibodies to high-risk outpatients recently diagnosed with COVID-19 reduced viral load and decreased the risk of hospitalization and death. Compared to bamlanivimab alone, use of the combination appears to be more effective in reducing viral load and less likely to result in development of treatment-emergent variants. How bamlanivimab and etesevimab compare with casirivimab (REGN10933) and imdevimab (REGN10987), two other monoclonal antibodies available for use together through an EUA, remains to be determined.
- FDA News Release. Coronavirus (COVID-19) update: FDA authorizes monoclonal antibodies for treatment of COVID-19. February 9, 2021. Available at: http://bit.ly/3d8grHG. Accessed Accessed March 18 2021.
- An EUA for bamlanivimab – a monoclonal antibody for COVID-19. Med Lett Drugs Ther 2020; 62:185.
- An EUA for casirivimab and imdevimab for COVID-19. Med Lett Drugs Ther 2020; 62:201.
- RL Gottlieb et al. Effect of bamlanivimab as monotherapy or in combination with etesevimab on viral load in patients with mild to moderate COVID-19: a randomized clinical trial. JAMA 2021; 325:632.
- FDA. Fact sheet for health care providers. Emergency Use Authorization (EUA) of bamlanivimab and etesevimab. Available at: https://bit.ly/3qfS6DN. Accessed January 6, 2022.
- Lilly News Release. Lilly's bamlanivimab and etesevimab together reduced hospitalizations and death in phase 3 trial for early COVID-19. March 10, 2021. Available at: http://bit.ly/2PRZmZ7. Accessed March 18, 2021.
- Lilly News Release. Lilly's bamlanivimab (LY-CoV555) administered with etesevimab (LY-CoV016) receives FDA emergency use authorization for COVID-19. February 9, 2021. Available at: http://bit.ly/3jGYIbM. Accessed March 18, 2021.