ISSUE1750
- Jean-Marie Pflomm, Pharm.D., Editor in Chief has disclosed no relevant financial relationships.
- Discuss the potential role of Lasix ONYU, a subcutaneous furosemide infusor, for treatment of edema in patients with heart failure.
The FDA has approved Lasix ONYU (SQ Innovation), a subcutaneous formulation of the loop diuretic furosemide administered via a wearable pump (on-body infusor), for treatment of edema in adults with chronic heart failure (HF). A similar product, Furoscix, is approved for treatment of edema in chronic HF or chronic kidney disease.1 Furosemide has been available for years in oral and IV formulations for such use.
DIURETICS IN HF — Loop diuretics such as furosemide, bumetanide, and torsemide are more effective than thiazide-type diuretics such as hydrochlorothiazide for treatment of edema. They are often administered intravenously to achieve rapid diuresis in patients with acute edema that is resistant to oral diuretics. IV loop diuretics are inexpensive, but they generally must be given by a healthcare professional.2
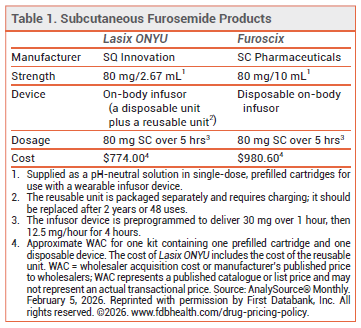
EFFICACY AND SAFETY — FDA approval of Lasix ONYU was based on the results of a trial demonstrating that a pH-neutral, subcutaneous formulation of furosemide infused by a conventional pump over 5 hours (30 mg over 1 hour, then 12.5 mg/hour for the next 4 hours) had an absolute bioavailability similar to that of IV furosemide (one 80-mg bolus dose over 2 minutes), and produced equivalent diuresis. When the same subcutaneous formulation of furosemide was administered via the on-body infusor, plasma concentrations of the drug and diuresis were similar to those achieved with the conventional pump. Adverse effects associated with the on-body infusor included administration-site pain and local skin reactions.3
DOSAGE AND ADMINISTRATION — Lasix ONYU is not intended for chronic use; patients should be switched to oral diuretic therapy as soon as possible. After assembly and placement on the abdomen using an adhesive patch, the infusor will deliver 80 mg of furosemide subcutaneously over 5 hours (30 mg over the first hour, followed by 12.5 mg per hour for the next 4 hours). A second 80-mg infusion may be administered in a 24-hour period if needed.
- In brief: Furoscix – a subcutaneous furosemide infusor for heart failure. Med Lett Drugs Ther 2023; 65:14.
- Drugs for chronic heart failure. Med Lett Drugs Ther 2025; 67:81.
- J Osmanska et al. A novel, small-volume subcutaneous furosemide formulation delivered by an abdominal patch infusor device in patients with heart failure: results of two phase I studies. Eur Heart J Cardiovasc Pharmacother 2024; 10:35. doi:10.1093/ehjcvp/pvad073
