ISSUE1560
Persons planning to travel outside the US should be up to date on routine vaccines and, depending on their destination, duration of travel, and planned activities, may also receive certain travel-specific vaccines. Tickborne encephalitis and dengue vaccines, which are not available in the US, are reviewed in a separate article available online. Detailed advice for travel to specific destinations is available from the Centers for Disease Control and Prevention (CDC) at www.cdc. gov/travel/destinations/list. Recommendations for administration of vaccines as part of routine adult immunization are discussed in a separate issue.1
TIMING OF VACCINE ADMINISTRATION
Multiple inactivated and/or live-attenuated vaccines can generally be given on the same day at different sites without decreasing antibody responses or increasing the risk of adverse effects. If two live-attenuated vaccines are not administered on the same day, the second one should generally be given at least one month after the first. An interrupted vaccine series does not have to be restarted; there is no maximum interval between doses of a primary series.2
Immunocompromised or pregnant patients generally should not receive live vaccines, although the benefit might outweigh the risk in some situations.3
VACCINES
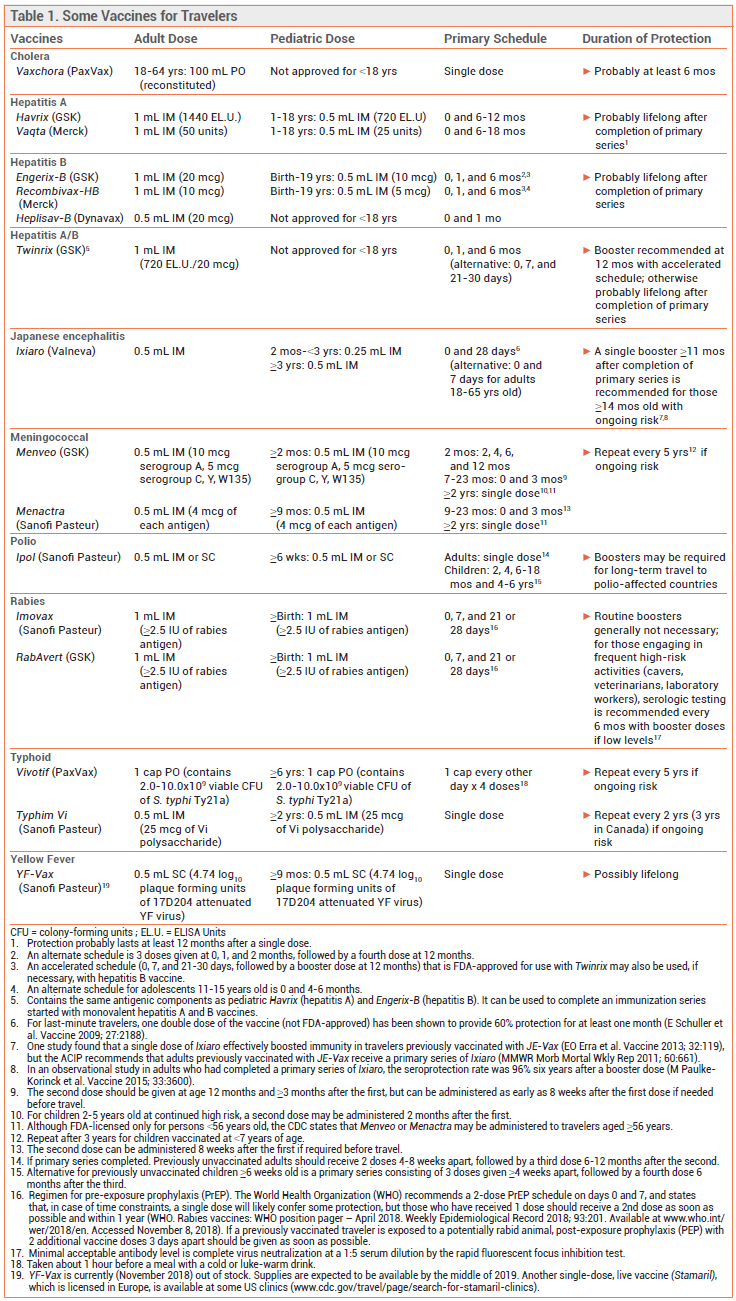
CHOLERA — Cholera is endemic in many countries in Africa and South and Southeast Asia. In recent years, most outbreaks have been caused by toxigenic strains of Vibrio cholerae serogroup O1. Cases caused by serogroup O139 have occurred in a few areas in Asia.
For most tourists, the risk of cholera is very low. Travelers who might have a higher risk of exposure include those who plan to work in refugee camps, in outbreak settings, or as healthcare providers, and those visiting friends and relatives or staying for extended periods in affected areas.
Recommendations – The US Advisory Committee on Immunization Practices (ACIP) recommends vaccination for persons 18-64 years old traveling to areas with endemic or epidemic cholera caused by toxigenic V. cholerae serogroup O1, including areas with cholera activity within the last year that are prone to recurrences of cholera epidemics.4
The Vaccine – Vaxchora, a single-dose, oral, live-attenuated cholera vaccine, is FDA-licensed to protect against disease caused by toxigenic V. cholerae serogroup O1 (but not serogroup O139 or other non-O1 serogroups) in adults 18-64 years old. It should be taken at least 10 days before potential cholera exposure.5
In a randomized trial, volunteers were challenged with wild-type V. cholerae serogroup O1 10 days or 3 months after vaccination; the seroconversion rate was 89% at 10 days and 90% at 180 days after vaccination. Vaccine efficacy against moderate or severe diarrhea was 90% at 10 days and 80% at 3 months.6
HEPATITIS A — Hepatitis A virus (HAV) is common in countries where sanitation is poor and access to clean water is limited, including many parts of Africa, Asia, Central and South America, and Eastern Europe. Areas with low levels of endemic HAV transmission include Western Europe, Japan, New Zealand, and Australia. HAV vaccination has been part of routine childhood immunization in the US since 2006.
Recommendations – HAV vaccine is recommended by the ACIP for all unvaccinated travelers going to countries with intermediate or high HAV endemicity.7 However, there is a potential risk of foodborne HAV infection even in countries with low endemicity; some experts advise all persons traveling outside the US to consider hepatitis A vaccination regardless of their destination.
The Vaccines – Monovalent HAV vaccines (Havrix, Vaqta) are usually administered in 2 IM doses at least 6 months apart. Antibodies reach protective levels 2-4 weeks after the first dose. Even when exposure to the virus occurs sooner than 4 weeks after vaccination, the traveler is usually protected because the hepatitis A incubation period is relatively long (average 28 days). Immunocompromised patients, who have reduced seroconversion rates after the first dose of HAV vaccine, may benefit from an extra priming dose; in a study in adults with drug-induced immunosuppression given 2 doses 4 weeks apart, the seroprotection rate at 2 months was 84%.8 A series started with one of the two monovalent hepatitis A vaccines may be completed with the other, or (in adults) with the combination hepatitis A/hepatitis B virus vaccine (Twinrix). A second dose given up to 11 years after the first has produced protective antibody levels.9
For older adults, immunocompromised patients, and those with chronic liver disease or other chronic medical conditions who will be traveling to an endemic area in ≤2 weeks, one dose of immune globulin (GamaSTAN S/D) should be given in addition to the initial dose of vaccine. Infants <6 months old and persons who elect not to receive the vaccine should receive a single dose of immune globulin before travel. Infants 6-11 months old may receive a single dose of vaccine before travel instead of immune globulin, followed by a complete 2-dose series beginning at ≥12 months of age; the rationale for off-label use of the vaccine in this age group is that HAV and live-attenuated measles, mumps, and rubella (MMR) vaccines can be administered simultaneously, but MMR vaccine cannot be administered with immune globulin.10 The recommended dosage of immune globulin, which was increased in 2017, varies according to the duration of travel: 0.1 mL/kg for up to one month of travel; 0.2 mL/kg for up to 2 months; 0.2 mL/kg every 2 months for longer travel.11
To view a more detailed table, click here.
HEPATITIS B — Hepatitis B surface antigen (HbsAg) prevalence is generally high in sub-Saharan Africa and the Western Pacific region and low in most countries in the Americas, Western Europe, Japan, and Australia.12 Hepatitis B virus (HBV) vaccine is part of routine childhood immunization in the US.
Recommendations – HBV vaccine is recommended by the ACIP for all travelers going to areas with intermediate or high prevalence of chronic HBV infection.9 The risk for most travelers is low. Travelers who engage in behaviors that increase the risk of transmission, such as injection drug use, unprotected sexual contact with new partners, dental treatment, skin perforation practices (tattoos, acupuncture, ear piercing), or medical tourism involving invasive medical treatment, should receive the HBV vaccine regardless of their destination.13
The Vaccines – Three monovalent HBV vaccines are available in the US (Engerix-B, Heplisav-B, Recombivax). Heplisav-B is a 2-dose vaccine that is only FDA-licensed for use in adults. The other vaccines are usually administered in 3 doses. In randomized clinical trials, seroprotection rates with 2 doses of Heplisav-B were significantly higher than those with 3 doses of Engerix-B.14
The ACIP recommends using the same vaccine for all doses of a hepatitis B series, but an interrupted series does not have to be restarted if the same vaccine is not available. Two doses of Heplisav-B can be used to complete a series started with a single dose of another monovalent vaccine or the combination HAV/HBV vaccine. One dose of Heplisav-B can be used as part of a 3-dose series.15
INFLUENZA — Influenza may be a risk in temperate areas of the Southern Hemisphere from April to September and of the Northern Hemisphere from October to March, and in the tropics year-round. Increased risk has been associated with travel in large tour groups and on cruise ships and airplanes, participation in mass gatherings, and staying abroad for >30 days. Persons traveling to East and Southeast Asia appear to have a higher risk of acquiring influenza than those traveling to other regions.16
Recommendations – The ACIP recommends routine annual vaccination against seasonal influenza for everyone ≥6 months old; detailed recommendations are included in a separate issue.17
Travelers not vaccinated during the preceding Northern Hemisphere influenza season should consider being vaccinated before going to the Southern Hemisphere during that region's influenza season or to the tropics in any season, or when traveling in a group with persons from the Southern Hemisphere during their influenza season.18
The Vaccines – Northern Hemisphere influenza vaccine is usually available in the US from August until the end of June. The vaccine formulations may be the same in the Northern and Southern Hemispheres. If they are different, high-risk patients from the Northern Hemisphere who travel to the Southern Hemisphere during that region's influenza season could consider vaccination prior to departure with a US-licensed Southern Hemisphere formulation (Fluzone Quadrivalent Southern Hemisphere), if available, or vaccination upon arrival in the Southern Hemisphere. Serum antibody levels peak about 2 weeks after vaccination in most adults.
JAPANESE ENCEPHALITIS — Japanese encephalitis is an uncommon but potentially fatal mosquito-borne viral disease that occurs in rural Asia, especially near pig farms and rice paddies. It is usually seasonal (May-October), but may occur year-round in tropical regions. The incidence in travelers has been very low.19
Recommendations – Vaccination is recommended for travelers who expect a long stay (≥1 month) in endemic areas or heavy exposure to mosquitoes (such as adventure travelers) during the transmission season. Vaccination should be considered for travelers spending <1 month in endemic areas during the transmission season if they will be sleeping without air conditioning, screens, or bed nets, or spending considerable time outside in rural or agricultural areas, especially in the evening or at night.20
The Vaccine – Ixiaro, an inactivated Vero cell culture-derived vaccine, is the only Japanese encephalitis vaccine currently available in the US.21 It is licensed by the FDA for use in adults and children ≥2 months old.22 Ixiaro is usually given in 2 doses 28 days apart. The FDA has now approved an accelerated schedule (0 and 7 days) of Ixiaro that can be used as an alternative for last-minute adult travelers ≤65 years old. In a randomized trial in healthy adults 18-65 years old, the accelerated schedule was noninferior to the standard schedule in terms of immunogenicity and safety at one month and one year after vaccination.23,24
In an observational study, 96% of adults who had completed a 2-dose primary series of Ixiaro and received one booster dose 15 months later were still seroprotected 6 years after their booster dose.25
A single dose of Ixiaro has been shown to effectively boost antibody levels in persons vaccinated with a previously available vaccine (JE-Vax), but the duration of protection is unknown; the ACIP recommends that persons previously vaccinated with JE-Vax who require continued protection receive a 2-dose primary series of Ixiaro.
MEASLES — Travel has been associated with importation of measles virus from other countries where it is endemic or large outbreaks are occurring, leading to outbreaks in the US, primarily among unvaccinated persons.26
Recommendations – The CDC recommends vaccination against measles for all persons without evidence of immunity, particularly international travelers. Children 6-11 months old should receive one MMR dose before traveling; they will still need 2 more doses of a measles-containing vaccine for routine immunization, beginning at ≥12 months of age. Previously unvaccinated children ≥12 months old, adolescents, and adults (born in 1957 or later [1970 in Canada]) should receive 2 doses of a measles-containing vaccine at least 28 days apart.27,28
The Vaccine – The measles vaccine is available as a live-attenuated vaccine in combination with mumps and rubella (MMR; M-M-R-II) or with mumps, rubella, and varicella (MMRV; ProQuad; approved only for children 12 months-12 years old).
MENINGOCOCCAL — Outbreaks of meningococcal disease can occur anywhere in the world, but they are most common in the African "meningitis belt" (semi-arid areas of sub-Saharan Africa extending from Senegal and Guinea eastward to Ethiopia). Recent outbreaks have primarily been due to Neisseria meningitidis serogroups C and W.
The Vaccines – Two quadrivalent inactivated vaccines against N. meningitidis serogroups A, C, W, and Y (MenACWY; Menactra; Menveo) are available in the US.29,30 Both contain meningococcal capsular polysaccharides conjugated to a protein carrier. A third unconjugated quadrivalent polysaccharide vaccine (Menomune) is no longer available in the US.
Persons traveling to Saudi Arabia during the Hajj or Umrah must present proof of immunization with MenACWY. The vaccine must be administered no fewer than 10 days and no more than 3 years before travel if the previously available polysaccharide vaccine was used and no more than 5 years before if the conjugate vaccine was used.31
Two vaccines against N. meningitidis serogroup B (MenB; Trumenba; Bexsero) are available for use in persons 10-25 years old. MenB vaccine is not routinely recommended for travel unless an outbreak has been reported.
Recommendations – Vaccination with MenACWY is recommended for travelers going anywhere in the meningitis belt during the dry season (December to June), and should also be considered for areas where outbreaks of N. meningitidis are occurring, particularly for travelers who will have prolonged contact with the local population, such as those living in a dormitory, military institution, or refugee camp, or working in a healthcare setting.28
PNEUMOCOCCAL — The incidence of pneumococcal disease is higher in some developing countries than in the US. Pneumococcal vaccination has been part of routine childhood immunization in the US since 2010.
The Vaccines – Two pneumococcal vaccines are available in the US: PCV13 (Prevnar 13), a conjugate vaccine that contains 13 serotypes of pneumococcus, and PPSV23 (Pneumovax 23), a 23-valent pneumococcal polysaccharide vaccine.
Recommendations – Travelers should be up to date on pneumococcal vaccination. It is recommended for healthy adults ≥65 years old and for some persons <65 years old with certain immunocompromising conditions or other risk factors (e.g., smoking and chronic diseases such as diabetes). Healthy adults ≥65 years old who have not previously received either vaccine should receive PCV13 first, followed at least one year later by PPSV23. A shorter interval between doses (PCV13 followed ≥8 weeks later by PPSV23) is recommended by the ACIP for adults with immunocompromising conditions and may be appropriate for some travelers with upcoming trips.1,32
POLIO — In 2018, the only countries with wild poliovirus circulation during the last 12 months were Pakistan and Afghanistan. Outbreaks of vaccine-derived poliovirus have occurred in some other countries, mainly in Africa.
Recommendations – All persons traveling to countries with wild or vaccine-derived poliovirus circulation should complete a primary series of inactivated polio vaccine (IPV; Ipol) before leaving the US. Adults who have not previously been immunized against polio should receive a 3-dose primary series of IPV (2 doses 4-8 weeks apart; third dose 6-12 months after the second). If protection is needed sooner, 2 or 3 doses can be given ≥4 weeks apart; if <4 weeks are available before protection is needed, a single dose is recommended. Adults who previously completed a primary series and have never had a booster should receive a single booster dose of IPV. Previously unimmunized children should receive a 4-dose primary series of IPV beginning at age ≥6 weeks; the first 3 doses should be given ≥4 weeks apart, followed by a fourth dose ≥6 months after the third. A child who received 4 doses before age 4 should be given a fifth dose at age 4-6 years.
Travelers planning to stay for >4 weeks in a country with recent wild or vaccine-derived poliovirus circulation may be required to present proof of polio vaccination when departing that country. Polio vaccination of travelers should be documented on an International Certificate of Vaccination or Prophylaxis (ICVP; "yellow card"). Children and adults who will be in a polio-infected country for >4 weeks, and whose last dose of polio vaccine was administered >12 months before the date they will be leaving that country, should receive an additional dose of IPV before leaving the US. Those who plan to reside in a polio-infected country for >12 months may be required to receive a dose of the polio vaccine that is available in that country (either IPV or oral polio vaccine) between 4 weeks and 12 months before their departure from the polio-infected country.33
RABIES — Rabies is highly endemic in many parts of Africa, Asia (particularly India, Pakistan, Bangladesh, and Bali, Indonesia), and Central and South America, but the risk to travelers is generally low.34
Recommendations – Pre-exposure vaccination (PrEP) against rabies is recommended for travelers with an occupational risk of exposure, for those (especially children) visiting endemic areas where immediate access to medical treatment, particularly rabies immune globulin (RIG), tends to be limited, and for outdoor-adventure travelers.35
After exposure to a potentially rabid animal, persons who received PrEP should promptly receive 2 additional doses of vaccine 3 days apart. For those who did not receive PrEP, the ACIP recommends human RIG plus 4 doses of vaccine given over 14 days; immunosuppressed patients should receive 5 doses of vaccine over 28 days.36 In one retrospective study, some patients did not develop adequate antibody levels after 4 doses of vaccine.37
The Vaccines – Two rabies antigen vaccines are available in the US (Imovax; RabAvert). According to the CDC, cell culture rabies vaccines available outside the US are acceptable alternatives to FDA-approved vaccines, but neural tissue vaccines, which have high rates of serious adverse effects, are not. RIG is not readily available in developing countries, and even when it is, its purity and potency may be less reliable. Purified equine-derived RIG is available in some developing countries and has been used effectively, with a low incidence of adverse effects.
TETANUS, DIPHTHERIA, AND PERTUSSIS — Vaccination against tetanus diphtheria, and pertussis, which is part of routine childhood immunization in the US, is recommended for all travelers.
Recommendations – Previously unimmunized children should receive 3 or (preferably) 4 doses of pediatric diphtheria, tetanus, and acellular pertussis vaccine (DTaP) before travel. An accelerated schedule can be used beginning at age ≥6 weeks: the first 3 doses are given 4 weeks apart and the fourth dose is given at age ≥12 months and ≥6 months after the third.
Adults with an uncertain history of primary vaccination should receive 3 doses of an inactivated tetanus and diphtheria toxoid (Td) vaccine. The first 2 doses should be administered ≥4 weeks apart and the third 6-12 months after the second. One of the 3 doses (preferably the first) should contain protein components of acellular pertussis combined with diphtheria and tetanus toxoids (Tdap) to also provide protection against pertussis. Two Tdap vaccines (Adacel; Boostrix) are available for use in adults, including those ≥65 years old.38
A booster dose of inactivated Td vaccine is recommended every 10 years for adults. All persons ≥11 years old who have completed a primary childhood series and have not yet received Tdap vaccine should receive a single dose, which can be given regardless of the interval since the last Td dose to provide pertussis protection before travel. Pregnant women should receive Tdap during each pregnancy.
Tetanus and diphtheria-containing vaccines may interact with carrier proteins on conjugate vaccines, suppressing the immune response to these vaccines; Tdap administration should be deferred until after administration of a meningococcal or pneumococcal conjugate vaccine.39
TYPHOID — Typhoid fever is usually acquired through consumption of food or water contaminated with Salmonella typhi. The risk is highest in South Asia. Other risk areas are East and Southeast Asia, Central and South America, the Caribbean, and Africa.
Recommendations – Vaccination is recommended for travelers going to areas where there is an increased risk of typhoid fever, especially if they expect a long stay or will be visiting friends or relatives or traveling outside routine tourist destinations.40,41
The Vaccines – A live-attenuated oral typhoid vaccine (Vivotif) is available for adults and children ≥6 years old. It is taken every other day as a single capsule (at least 1 hour before eating) for a total of 4 doses, beginning at least 2 weeks before departure. The capsules must be refrigerated. Antibiotics should be avoided for at least 24 hours before administration of the first dose and for 1 week after administration of the last dose. The vaccine provides protection for about 5 years.
A purified capsular polysaccharide parenteral vaccine (Typhim Vi) is FDA-licensed for use in adults and children ≥2 years old. It is given at least 2 weeks before departure and provides protection for about 2 years.
A combined hepatitis A/typhoid vaccine (Vivaxim) is available in Canada.
VARICELLA — Varicella is endemic worldwide. Vaccination against varicella has been part of routine childhood immunization in the US since 1996.
Recommendations – Persons born in the US before 1980 are generally considered immune to varicella.1 Vaccination is recommended for all travelers ≥12 months old without evidence of immunity. Before traveling abroad, children 1-3 years old should receive 1 dose of vaccine and persons ≥4 years old should receive 2 doses. The minimum interval between doses should be 3 months for persons <13 years old and 4 weeks for those ≥13 years old.28
The Vaccines – Two varicella-containing vaccines are available in the US: single-antigen varicella vaccine (Varivax), which is licensed for use in persons ≥12 months old, and combination measles-mumps-rubella-varicella vaccine (MMRV; ProQuad), which is only licensed for use in children 12 months-12 years old.
YELLOW FEVER — Yellow fever virus is transmitted by the bite of an infected mosquito. It is endemic in tropical South America and sub-Saharan Africa.
Recommendations – The ACIP recommends administration of a single dose of yellow fever vaccine at least 10 days before travel to endemic areas.42 Booster doses are no longer recommended for most travelers because a single dose has been shown to provide protection for many decades. Additional doses are still recommended for certain populations: women who were pregnant when they received their initial dose should receive one additional dose before traveling to an endemic area; persons who underwent a hematopoietic stem cell transplantation after being vaccinated should be revaccinated before traveling if they are sufficiently immunocompetent; persons who were infected with HIV when they received their last dose should receive a booster dose every 10 years if they continue to be at risk. A booster dose given at least 10 years after the previous dose may also be considered for travelers at increased risk of exposure, such as those planning prolonged travel in an endemic area or those traveling to highly endemic areas during peak transmission season or to an area with an ongoing outbreak.43
Some countries require an International Certificate of Vaccination or Prophylaxis (ICVP, "yellow card") as proof of vaccination against yellow fever, or a physician's waiver letter, from all entering travelers; other countries require evidence of vaccination from travelers coming from or traveling through endemic or infected areas, including brief airport transits. An updated list of countries requiring proof of yellow fever vaccination is available at www.cdc.gov/travel.
The Vaccine – US-licensed yellow fever vaccine (YF-Vax) is a live-attenuated vaccine prepared in eggs. It is usually available from providers certified by state health departments, but at press time it is temporarily out of stock; supplies are expected to be available by the middle of 2019. Some clinics are offering an alternative single-dose live vaccine (Stamaril – Sanofi Pasteur). A list of these clinics is available at wwwnc.cdc.gov/travel/page/search-for-stamaril-clinics. Stamaril, which has been used for decades in Europe, is available in the US under an investigational new drug program. It is comparable in efficacy and safety to YF-Vax.
Yellow fever vaccine-associated viscerotropic disease, a severe systemic illness that can cause fatal organ failure, has been reported rarely. It has occurred only in first-time recipients and is most common in persons >60 years old. Vaccine-associated neurologic disease (encephalitis, Guillain-Barré syndrome, Bell's palsy) has also occurred almost exclusively in first-time recipients; the risk is increased in infants and persons >60 years old.44,45
Caution is advised for vaccination of infants 6-8 months old, first-time recipients ≥60 years old, and those with asymptomatic HIV infection and moderate immune suppression (CD4 counts 200-499 cells/mm3). In a small study, relapse rates were significantly higher among travelers with multiple sclerosis who received yellow fever vaccine.46 There have been case reports of vaccine-associated neurologic disease in breastfed infants of recently vaccinated women.47 The vaccine is contraindicated in infants <6 months old, in persons with immunodeficiencies or receiving immunosuppressive or immunomodulatory therapies, and in those with HIV infection and CD4 counts <200 cells/mm3.42
View the Expanded Table: Some Vaccines for Travelers
- Adult immunization. Med Lett Drugs Ther 2018; 60:73.
- DO Freedman et al. Medical considerations before international travel. N Engl J Med 2016; 375:247.
- AT Kroger et al. General best practice guidelines for immunization. Best practices guidance of the Advisory Committee on Immunization Practices (ACIP). Available at: https://www.cdc.gov. Accessed November 8, 2018.
- KK Wong et al. Recommendations of the Advisory Committee on Immunization Practices for use of cholera vaccine. MMWR Morb Mortal Wkly Rep 2017; 66:482.
- An oral cholera vaccine for travelers (Vaxchora). Med Lett Drugs Ther 2016; 58:113.
- WH Chen et al. Single-dose live oral cholera vaccine CVD 103-HgR protects against human experimental infection with Vibrio cholerae 01 El Tor. Clin Infect Dis 2016; 62:1329.
- KH Jacobsen. Globalization and the changing epidemiology of hepatitis A virus. Cold Spring Harb Perspect Med 2018; 8(10).
- A Rosdahl et al. An extra priming dose of hepatitis A vaccine to adult patients with rheumatoid arthritis and drug induced immunosuppression – a prospective, open-label, multi-center study. Travel Med Infect Dis 2018; 21:43.
- JJ Ott and ST Wiersma. Single-dose administration of inactivated hepatitis A vaccination in the context of hepatitis A vaccine recommendations. Int J Infect Dis 2013; 17:e939.
- Centers for Disease Control and Prevention. Advisory Committee on Immunization Practices (ACIP) summary report, February 21-22, 2018, Atlanta, Georgia. Available at: www.cdc.gov. Accessed November 8, 2018.
- NP Nelson. Updated dosing instructions for immune globulin (human) GamaSTAN S/D for hepatitis A virus prophylaxis. MMWR Morb Mortal Wkly Rep 2017; 66:959.
- A Schweitzer et al. Estimations of worldwide prevalence of chronic hepatitis B virus infection: a systematic review of data published between 1965 and 2013. Lancet 2015; 386:1546.
- DF Johnson et al. Hepatitis B and C infection in international travelers. J Travel Med 2013; 20:194.
- A two-dose hepatitis B vaccine for adults (Heplisav-B). Med Lett Drugs Ther 2018; 60:17.
- S Schillie et al. Prevention of hepatitis B virus infection in the United States: recommendations of the Advisory Committee on Immunization Practices. MMWR Recomm Rep 2018; 67(1):1.
- M Goeijenbier et al. Travellers and influenza: risks and prevention. J Travel Med 2016; 24(1).
- Influenza vaccine for 2018-2019. Med Lett Drugs Ther 2018; 60:169.
- Centers for Disease Control and Prevention. Use of northern hemisphere influenza vaccines by travelers to the southern hemisphere. MMWR Morb Mortal Wkly Rep 2009; 58:312.
- SL Hills et al. Japanese encephalitis in travelers from non-endemic countries, 1973-2008. Am J Trop Med Hyg 2010; 82:930.
- E Mirzaian et al. Mosquito-borne illnesses in travelers: a review of risk and prevention. Pharmacotherapy 2010; 30:1031.
- A new Japanese encephalitis vaccine (Ixiaro). Med Lett Drugs Ther 2009; 51:66.
- Use of Japanese encephalitis vaccine in children: recommendations of the advisory committee on immunization practices, 2013. MMWR Morb Mortal Wkly Rep 2013; 62:898.
- T Jelinek et al. Short-term immunogenicity and safety of an accelerated pre-exposure prophylaxis regimen with Japanese encephalitis vaccine in combination with a rabies vaccine: a phase III multicenter, observer-blind study. J Travel Med 2015; 22:225.
- JP Cramer et al. One-year immunogenicity kinetics and safety of a purified chick embryo cell rabies vaccine and an inactivated Vero cell-derived Japanese encephalitis vaccine administered concomitantly according to a new, 1-week, accelerated primary series. J Travel Med 2016; 23(3).
- M Paulke-Korinek et al. Persistence of antibodies six years after booster vaccination with inactivated vaccine against Japanese encephalitis. Vaccine 2015; 33:3600.
- PA Gastañaduy et al. Measles - United States, January 1-May 23, 2014. MMWR Morbid Mortal Wkly Rep 2014; 63:496.
- HQ McLean et al. Prevention of measles, rubella, congenital rubella syndrome, and mumps, 2013: summary recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm Rep 2013; 62(RR-4):1.
- Centers for Disease Control and Prevention. CDC Yellow Book 2018: Health Information for International Travel. New York: Oxford University Press, 2017. Available at: https://wwwnc.cdc.gov. Accessed November 8, 2018.
- A new conjugate meningococcal vaccine (Menveo). Med Lett Drugs Ther 2010; 52:59.
- JR MacNeil et al. Use of MenACWY-CRM vaccine in children aged 2 through 23 months at increased risk for meningococcal disease: recommendations of the Advisory Committee on Immunization Practices, 2013. MMWR Morb Mortal Wkly Rep 2014; 63:527.
- AC Cohn et al. Prevention and control of meningococcal disease: recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm Rep 2013; 62(RR-2):1.
- M Kobayashi et al. Intervals between PCV13 and PPSV23 vaccines: recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Morb Mortal Wkly Rep 2015; 64:944.
- GS Wallace et al. Interim CDC guidance for polio vaccination for travel to and from countries affected by wild poliovirus. MMWR Morb Mortal Wkly Rep 2014; 63:591.
- C Malerczyk et al. Imported human rabies cases in Europe, the United States, and Japan, 1990 to 2010. J Travel Med 2011; 18:402.
- AJ Nigg and PL Walker. Overview, prevention, and treatment of rabies. Pharmacotherapy 2009; 29:1182.
- CE Rupprecht et al. Use of a reduced (4-dose) vaccine schedule for postexposure prophylaxis to prevent human rabies: recommendations of the Advisory Committee on Immunization Practices. MMWR Recomm Rep 2010; 59(RR-2):1.
- M Uwanyiligira et al. Rabies postexposure prophylaxis in routine practice in view of the new Centers for Disease Control and Prevention and World Health Organization recommendations. Clin Infect Dis 2012; 55:201.
- Prevention and treatment of pertussis. Med Lett Drugs Ther 2012; 54:73.
- M Tashani et al. Tetanus-diphtheria-pertussis vaccine may suppress the immune response to subsequent immunization with pneumococcal CRM197-conjugate vaccine (co-administered with quadrivalent meningococcal TT-conjugate vaccine): a randomized, controlled trial. J Travel Med 2017; 24(4).
- C Greenaway et al. Summary of the Statement on International Travellers and Typhoid by the Committee to Advise on Tropical Medicine and Travel (CATMAT). Can Commun Dis Rep 2014; 40:60.
- JA Whitaker et al. Rethinking typhoid fever vaccines: implications for travelers and people living in highly endemic areas. J Travel Med 2009; 16:46.
- JE Staples et al. Yellow fever vaccine: recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm Rep 2010; 59(RR-7):1.
- JE Staples et al. Yellow fever vaccine booster doses: recommendations of the Advisory Committee on Immunization Practices, 2015. MMWR Morb Mortal Wkly Rep 2015; 64:647.
- RE Thomas et al. How many published cases of serious adverse events after yellow fever vaccination meet Brighton Collaboration diagnostic criteria? Vaccine 2013; 31:6201.
- RE Thomas et al. The safety of yellow fever vaccine 17D or 17DD in children, pregnant women, HIV+ individuals, and older persons: systematic review. Am J Trop Med Hyg 2012; 86:359.
- MF Farez and J Correale. Yellow fever vaccination and increased relapse rate in travelers with multiple sclerosis. Arch Neurol 2011; 68:1267.
- S Kuhn et al. Case report: probable transmission of vaccine strain of yellow fever virus to an infant via breast milk. CMAJ 2011; 183:E243.