ISSUE1333
Aspirin and other nonsteroidal anti-inflammatory drugs (NSAIDs) are common causes of peptic ulcer disease. Patients infected with Helicobacter pylori who take aspirin or another NSAID have an especially high risk.1 Drugs that have been tried for prevention of ulcers in patients taking NSAIDs including H2-receptor antagonists, proton pump inhibitors (PPIs), aluminum- or magnesium-containing antacids, the prostaglandin misoprostol (Cytotec ,and others), and antibiotics to eradicate H. pylori.2
H2-RECEPTOR ANTAGONISTS — NSAIDs are more likely to cause gastric than duodenal ulcers. High doses of an H2-receptor antagonist have been shown to prevent NSAID-related gastric ulcers. In a 6-month study in patients on long-term NSAID therapy, famotidine 40 mg twice daily was significantly superior to placebo in preventing gastric ulcers found on endoscopy, which occurred in 20% of placebo-treated patients and 8% of those on famotidine.3 In a 12-week randomized, double-blind trial comparing famotidine 20 mg twice daily to placebo in 404 adult patients taking aspirin 75-325 mg daily with or without clopidogrel (Plavix) or dipyridamole (Persantine, and others), the prevalence of gastric ulcers found on endoscopy was 15% with placebo and 3.4% with famotidine, and that of duodenal ulcers was 8.5% and 0.5%, respectively.4 However, continued use of these agents leads to pharmacologic tolerance and loss of effectiveness over time.2
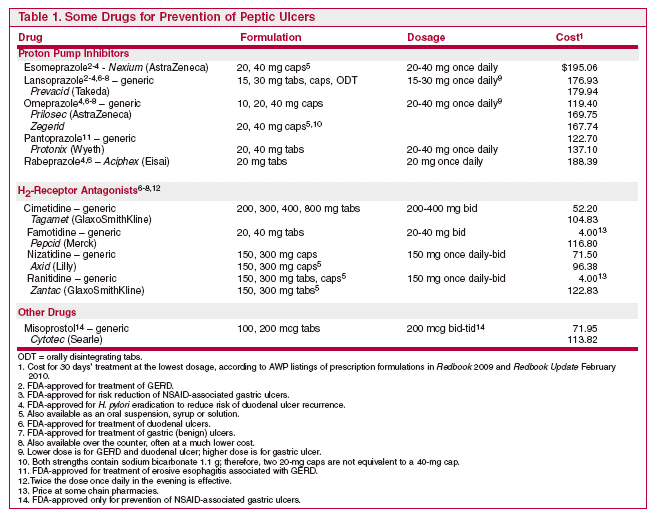
PROTON PUMP INHIBITORS (PPIs) — A randomized, double-blind trial in 169 patients taking NSAIDs compared omeprazole 20 mg once daily with placebo. After 6 months, the incidence of ulcers seen at endoscopy was 3.6% in the patients on omeprazole and 16.5% in those on placebo.5 A more recent 2-year study (VENUS) found that esomeprazole 20 or 40 mg once daily for 6 months was superior to placebo in preventing ulcers in NSAID users who had additional risk factors such as advanced age or a past history of ulcer; among 844 such patients, endoscopic ulcers developed in 20% with placebo, 5% with esomeprazole 20 mg, and 4% with esomeprazole 40 mg.6 Another trial compared esomeprazole 20 or 40 mg once daily with placebo in patients who were taking aspirin 75-325 mg daily and had one or more additional risk factors for ulceration, such as age over 65 or a past history of peptic ulcer disease; after 26 weeks, endoscopic ulcers were present in 7.4% of placebo-treated patients and in 1.1% and 1.5% of those on esomeprazole 20 or 40 mg, respectively.7
PPIs have been shown to be more effective than H2-receptor antagonists in prevention of NSAID-related ulcers and at least as effective as misoprostol.8,9
MISOPROSTOL — Misoprostol (Cytotec, and others), a synthetic prostaglandin E1 analog, can prevent gastric and duodenal ulcers in patients on chronic NSAID therapy. It may be as effective as a PPI, but requires multiple daily dosing and is not as well tolerated. Abdominal pain and dose-related diarrhea are the most common adverse effects of misoprostol. Nausea can also occur. Misoprostol is an abortifacient and is contraindicated during pregnancy.10
H. PYLORI ERADICATION — A meta-analysis of five prospective trials of H. pylori eradication showed a statistically significant reduction in the risk of endoscopic ulcers in patients who had not yet begun NSAID treatment, but not among those already taking an NSAID.11 The American College of Gastroenterology now recommends considering routine testing for H. pylori before starting long-term therapy with an NSAID.1
ANTACIDS — There is no convincing evidence that long-term use of aluminum- or magnesium-containing antacids can prevent development of peptic ulcers in patients taking aspirin or NSAIDs. Other problems with antacid use are the requirements for multiple daily doses and adverse effects on bowel habits: constipation with aluminum-containing products and diarrhea with magnesium.
CONCLUSION — Taking a proton pump inhibitor can prevent aspirin- or NSAID-associated ulcers detected on endoscopy. To what extent it prevents clinical symptoms or bleeding remains to be determined.
1. FL Lanza et al. Guidelines for prevention of NSAID-related ulcer complications. Am J Gastroenterol 2009; 104:728.
2. Treatment of peptic ulcers and GERD. Treat Guidel Med Lett 2008; 6:55.
7. JM Scheiman et al. Prevention of low-dose acetylsalicylic acidassociated gastric/duodenal ulcers with esomeprazole 20 mg and 40 mg once daily in patients at increased risk of ulcer development: a randomized controlled trial (OBERON). Gastroenterology 2009; 136:A-70 (abstract 412).
