ISSUE1674
- Mark Abramowicz, M.D., President has disclosed no relevant financial relationships.
- Jean-Marie Pflomm, Pharm.D., Editor in Chief has disclosed no relevant financial relationships.
- Brinda M. Shah, Pharm.D., Consulting Editor has disclosed no relevant financial relationships.
- Explain the current approach to the outpatient management of common bacterial respiratory tract infections in adults and children.
- Discuss the pharmacologic agents available for treatment of common bacterial respiratory tract infections and compare them based on their efficacy, dosage and administration, and potential adverse effects.
- Determine the most appropriate therapy given the clinical presentation of an individual patient with a common bacterial respiratory tract infection.
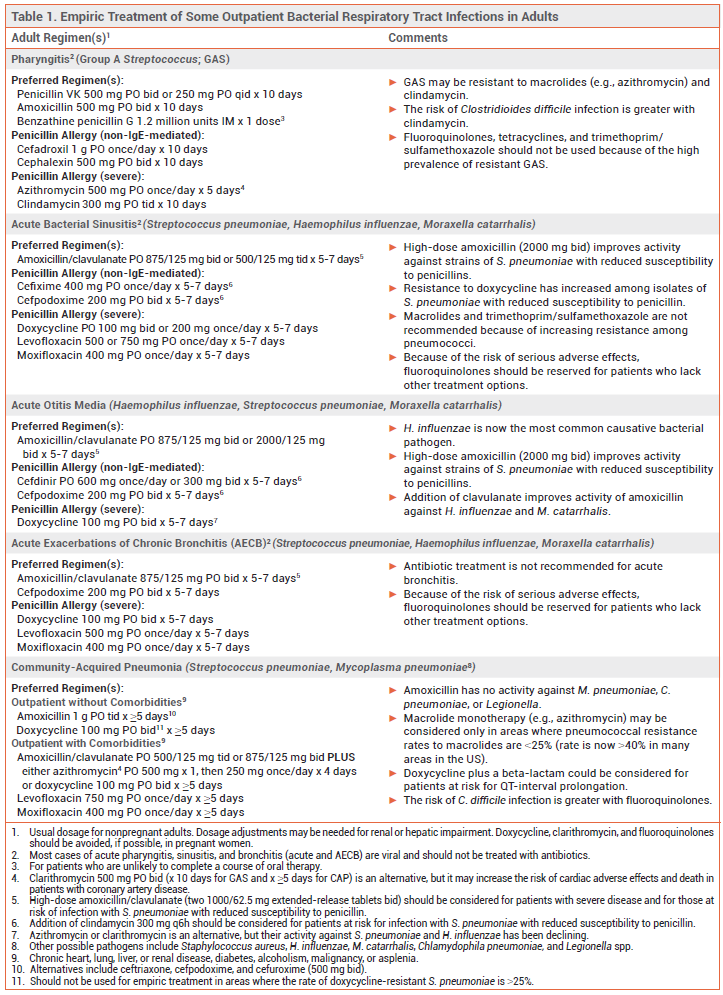
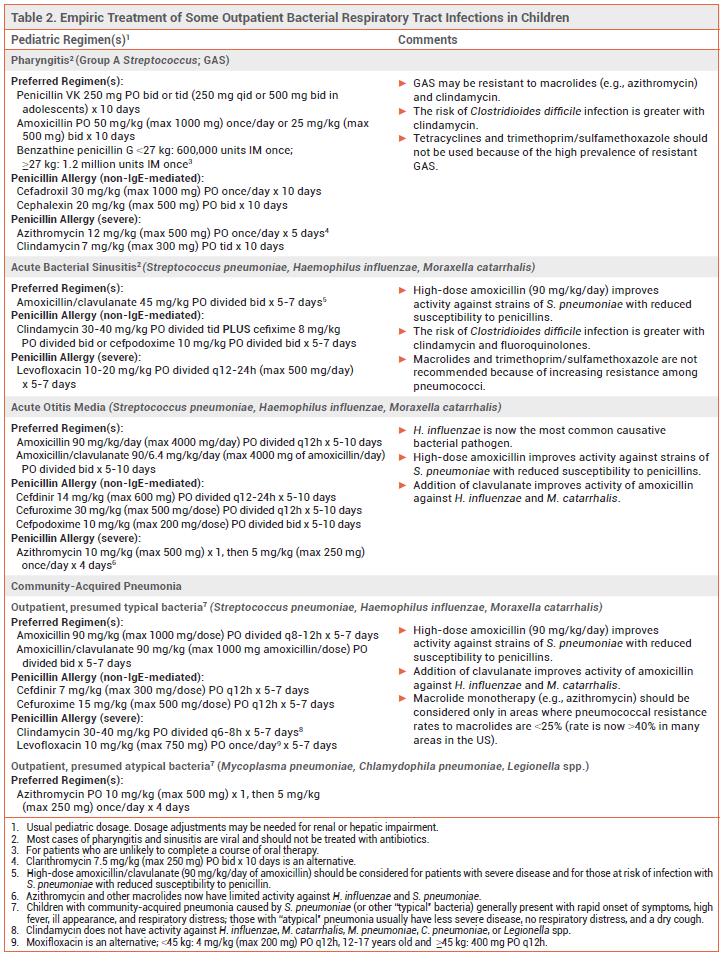
Most respiratory tract infections are caused by viruses. Bacterial respiratory tract infections are usually treated empirically with antibiotic therapy that targets the most probable causative pathogens. Recommended antibiotic regimens for outpatient treatment of some common respiratory tract infections are listed in Table 1 for adults and Table 2 for children.
GROUP A STREPTOCOCCAL PHARYNGITIS — Most cases of acute pharyngitis are caused by viruses, such as influenza, SARS-CoV-2, Epstein-Barr, rhinovirus, and HIV, and should not be treated with antibiotics. Group A Streptococcus (GAS; Streptococcus pyogenes) is the most common bacterial pathogen.
Diagnosis – GAS pharyngitis should be diagnosed by nucleic acid amplification test or rapid antigen detection test; use of history and physical examination alone is unreliable. In adults, throat culture can be considered if a rapid antigen test is negative and clinical presentation suggests bacterial infection. In children and adolescents, throat culture is always recommended when a rapid antigen test is negative and GAS pharyngitis is suspected. Follow-up culture is not needed after a negative nucleic acid amplification test.
Standard Treatment – Patients who test positive for GAS should be treated with a 10-day course of penicillin or amoxicillin.1 Those with a non-IgE-mediated allergy to penicillin can be treated with a first-generation cephalosporin such as cephalexin; the risk of cross-reactivity with penicillins and cephalosporins is low. Clindamycin or a macrolide such as azithromycin can be used in patients with a severe penicillin allergy, but >20% of GAS pharyngeal isolates may be resistant to these drugs.2,3 Fluoroquinolones, tetracyclines, and trimethoprim/sulfamethoxazole should not be used for treatment of GAS pharyngitis because of the high prevalence of resistance.
SINUSITIS — Acute sinusitis is usually caused by a viral infection and should not be treated with antibiotics. Symptoms can be managed with analgesics, an intranasal corticosteroid, steam inhalation, and/or sterile nasal saline irrigation. Bacterial sinusitis should be suspected in patients with a high fever and symptoms that are severe, persist for ≥10 days, or recur following resolution of a viral upper respiratory tract infection. Potential pathogens include Streptococcus pneumoniae, Haemophilus influenzae, and Moraxella catarrhalis. Bacterial sinusitis in otherwise healthy adults often improves without antibiotic therapy.4,5
Standard Treatment – Amoxicillin/clavulanate is the antibiotic of choice for empiric treatment of acute bacterial sinusitis in adults and children5,6; addition of clavulanate to amoxicillin improves its activity against beta-lactamase-producing organisms (e.g., H. influenzae, M. catarrhalis). A high dose of the amoxicillin component can be considered for patients at increased risk of resistant S. pneumoniae (e.g., ≥65 years old, comorbidities, immunosuppressed).
Doxycycline is an option for adults with a penicillin allergy, but resistance to doxycycline has increased, particularly among isolates of S. pneumoniae with reduced susceptibility to penicillin. Levofloxacin or moxifloxacin can also be used, but the FDA has warned that the risk of rare but serious adverse effects with fluoroquinolones, including tendinitis and tendon rupture, peripheral neuropathy, CNS effects, and QT-interval prolongation, generally outweighs their benefits for treatment of uncomplicated infections such as sinusitis.7 A third-generation oral cephalosporin (e.g., cefpodoxime) with or without clindamycin is an alternative for patients with a non-IgE-mediated penicillin allergy; the risk of cross-reactivity between penicillins and later-generation cephalosporins is very low.4-6,8
Macrolides (e.g., azithromycin) and trimethoprim/sulfamethoxazole are not recommended for treatment of acute sinusitis because of increasing resistance among S. pneumoniae.4
ACUTE OTITIS MEDIA — Acute otitis media (AOM) is frequently associated with viral respiratory tract infections. It is most common in children. S. pneumoniae and H. influenzae are the most common bacterial pathogens. Since the introduction of routine pneumococcal vaccination in the US, the prevalence of S. pneumoniae has declined, but the pneumococcal serotypes that do cause AOM often have reduced susceptibility to amoxicillin.9,10 H. influenzae that produce beta-lactamase and are resistant to amoxicillin are now a common cause of AOM in both children and adults.9 M. catarrhalis, which also produces beta-lactamase and is resistant to amoxicillin, is another frequently isolated bacterial pathogen in children with AOM.9,10 Staphylococcus aureus is a potential pathogen in adults.
Standard Treatment – Antimicrobial therapy can shorten the duration of AOM symptoms and prevent complications, but severe symptoms generally last only a few days and most infections in children resolve without treatment. Children <2 years old and those with severe infection (i.e., otorrhea, severe or persistent otalgia for >48 hours, or a temperature ≥102.2°F) should receive immediate antibiotic treatment. In children ≥2 years old with less severe symptoms, watchful waiting combined with analgesia is appropriate; if symptoms fail to improve within 48-72 hours, antibiotic therapy should be started. Adults with AOM should be treated with antibiotics.
Guidelines for treatment of AOM in children, which were last published in 2013, recommended high-dose amoxicillin (90 mg/kg/day) for initial treatment in most children. High doses are used to improve activity against less susceptible strains of S. pneumoniae.11
To treat beta-lactamase-producing H. influenzae, many expert clinicians now recommend standard- or high-dose amoxicillin/clavulanate for first-line empiric treatment of AOM in all children without a penicillin allergy.10,12 Amoxicillin/clavulanate is the antibiotic of choice for adults; a high dose (2000/125 mg bid) is recommended for those who are at increased risk of infection with a resistant strain of S. pneumoniae (e.g., recent antibiotic use or hospitalization, ≥65 years old, immunocompromised).
A second- or third-generation cephalosporin (e.g., oral cefdinir, cefpodoxime, or cefuroxime, IM/IV ceftriaxone) can be used in adults or children with a history of non-IgE-mediated penicillin allergy; the risk of cross-reactivity between penicillins and later-generation cephalosporins is very low.8
Macrolides (e.g., azithromycin) now have limited activity against S. pneumoniae and H. influenzae; they should be considered only for patients with a history of serious penicillin allergy. Clindamycin may be an alternative for treatment of AOM caused by S. pneumoniae in patients with a serious penicillin allergy; it has no activity against H. influenzae or M. catarrhalis.
BRONCHITIS — Acute bronchitis in otherwise healthy persons is usually viral. Symptoms are caused by inflammation and are self-limited. Antibiotic treatment is not recommended.6
Acute exacerbations of chronic bronchitis (AECB) are also often viral in origin, but moderate to severe exacerbations in patients with COPD are usually treated with antibiotics.13 As with sinusitis, bacterial AECBs are often caused by S. pneumoniae, H. influenzae, or M. catarrhalis. Amoxicillin/clavulanate is the treatment of choice. Doxycycline can be considered for patients who are allergic to penicillins. Because of the risk of serious adverse effects, a fluoroquinolone (levofloxacin or moxifloxacin) should be reserved for patients who lack other treatment options.7
COMMUNITY-ACQUIRED PNEUMONIA — Viruses are common causes of community-acquired pneumonia (CAP). The most common bacterial pathogens responsible for CAP in adults include S. pneumoniae, H. influenzae, M. catarrhalis, and Mycoplasma pneumoniae. In children ≥5 years old, S. pneumoniae and M. pneumoniae are most common. Pneumococcal vaccination has substantially reduced the incidence of CAP caused by S. pneumoniae in the US.14
Methicillin-resistant S. aureus (MRSA) and resistant gram-negative bacteria can sometimes cause CAP. Patients with previous respiratory isolation of one of these pathogens, a recent viral infection, or recent hospitalization with parenteral antibiotic treatment are at increased risk.15
Standard Treatment of Outpatient CAP – For many years, monotherapy with an oral macrolide such as azithromycin was a regimen of choice for empiric outpatient treatment of CAP in otherwise healthy adults without comorbidities (chronic heart, lung, liver or renal disease, diabetes, alcoholism, malignancy, or asplenia). The incidence of macrolide-resistant S. pneumoniae is now >40% in many areas in the US, however, and current guidelines recommend that oral macrolide monotherapy be considered only in areas where pneumococcal resistance rates to macrolides are <25%. High-dose amoxicillin (1 g tid) or doxycycline is now recommended for empiric treatment of CAP in most cases.15 High-dose amoxicillin monotherapy provides coverage against S. pneumoniae, but it has no activity against M. pneumoniae, Chlamydophila pneumoniae, or Legionella spp.; some experts would add a macrolide or doxycycline to cover these pathogens.
For outpatient treatment of CAP in adults with comorbidities (chronic heart, lung, liver or renal disease, diabetes, alcoholism, malignancy, or asplenia), an oral beta-lactam (amoxicillin/clavulanate, cefpodoxime, or cefuroxime) and either a macrolide (azithromycin, clarithromycin) or doxycycline is recommended. Monotherapy with an oral fluoroquinolone (levofloxacin or moxifloxacin) is an alternative.15
Macrolides and fluoroquinolones can prolong the QT interval and rarely cause life-threatening ventricular arrhythmias; these drugs should be used with caution in patients with cardiovascular disease or risk factors for QT-interval prolongation and arrhythmias.16 Although clinical data are limited, some expert clinicians would use doxycycline plus a beta-lactam in such patients.
Children with CAP caused by S. pneumoniae (or other "typical" bacteria) generally present with rapid symptom onset, high fever, ill appearance, and respiratory distress. Those with CAP due to an "atypical" pathogen (e.g., M. pneumoniae) usually have less severe disease, a dry cough, and no respiratory distress. High-dose amoxicillin (90 mg/kg/day) is recommended for treatment of CAP presumed to be caused by typical bacteria in children; high-dose amoxicillin/clavulanate is an alternative. In children with suspected M. pneumoniae infection, macrolide monotherapy is recommended. Possible alternatives include doxycycline or a fluoroquinolone (levofloxacin or moxifloxacin).17
Duration of Antimicrobial Therapy – Antibiotic treatment for CAP should be continued until clinical stability is achieved (usually within 48-72 hours) and for at least 5 days. Short courses of treatment (5-7 days) appear to be similar in efficacy to longer courses (8-10 days).18
Newer Antibiotics – Data supporting the efficacy of newer FDA-approved antibiotics for treatment of CAP, including the fluoroquinolone delafloxacin (Baxdela),19 the tetracycline omadacycline (Nuzyra),20 and the pleuromutilin lefamulin (Xenleta)21 are limited. Until more data become available, empiric regimens with a longer record of efficacy and safety are preferred.
Adjunctive Corticosteroids – There are no data supporting the use of adjunctive corticosteroids for treatment of mild to moderate CAP.22
ADVERSE EFFECTS — Beta-lactam antibiotics (penicillins and cephalosporins) can cause rash, diarrhea, nausea, vomiting, allergic reactions, hemolytic anemia, neutropenia, cholestatic hepatitis, serum sickness, and seizures. Amoxicillin/clavulanate and cephalosporin antibiotics cause a higher incidence of diarrhea than amoxicillin.
Doxycycline can cause GI adverse effects and photosensitivity.
Azithromycin and clarithromycin can cause GI adverse effects, headache, dizziness, vaginitis, and QT-interval prolongation. Clarithromycin can also cause dysgeusia and hepatic enzyme elevations. The FDA has warned that use of clarithromycin may increase the risk of cardiovascular morbidity and mortality in patients with heart disease.23
Use of fluoroquinolones has been associated with GI adverse effects, tremors, rash, oral and vaginal Candida infections, eosinophilia, neutropenia, leukopenia, increased aminotransferase and serum creatinine levels, insomnia, photosensitivity reactions, and peripheral neuropathy. They have also been reported to cause hyperglycemia and severe hypoglycemia, especially in older adults and patients with diabetes. Central nervous system effects, including seizures, delirium, agitation, nervousness, and disturbances in attention, memory, and orientation, have occurred. Other serious adverse effects include tendinitis, tendon rupture, aortic aneurysm, exacerbation of myasthenia gravis, Clostridioides difficile infection, and QT-interval prolongation (except delafloxacin19) and torsades de pointes.24
Clindamycin causes GI adverse effects and is associated with an increased risk of C. difficile infection. Skin rash is common and other allergic reactions can occur.
DRUG INTERACTIONS — Coadministration of antacids or products containing aluminum, calcium, magnesium, or iron can decrease absorption of doxycycline and fluoroquinolones. Administration should be separated by several hours.
Concurrent use of azithromycin, clarithromycin, or fluoroquinolones with other QT-interval-prolonging drugs can result in additive effects.16
Use of fluoroquinolones with antihyperglycemic drugs may increase the risk of hypoglycemia. Concurrent use of fluoroquinolones and nonsteroidal anti-inflammatory drugs (NSAIDs) may lower the seizure threshold.
PREGNANCY — Doxycycline, clarithromycin, and fluoroquinolones should be avoided if possible in pregnant women.
- ST Shulman et al. Clinical practice guideline for the diagnosis and management of group A streptococcal pharyngitis: 2012 update by the Infectious Diseases Society of America. Clin Infect Dis 2012; 55:1279. doi:10.1093/cid/cis847
- GP DeMuri et al. Macrolide and clindamycin resistance in group A streptococci isolated from children with pharyngitis. Pediatr Infect Dis J 2017; 36:342. doi:10.1097/inf.0000000000001442
- CDC. Antibiotic resistance threats in the United States, 2019. Atlanta, GA: U.S. Department of Health and Human Services, CDC; 2019. Available at: https://bit.ly/3LTYOwM. Accessed March 30, 2023.
- AW Chow et al. IDSA clinical practice guideline for acute bacterial rhinosinusitis in children and adults. Clin Infect Dis 2012; 54:e72. doi:10.1093/cid/cir1043
- RM Rosenfeld et al. Clinical practice guideline (update): adult sinusitis executive summary. Otolaryngol Head Neck Surg 2015; 152:598. doi:10.1177/0194599815574247
- AM Harris et al. Appropriate antibiotic use for acute respiratory tract infection in adults: advice for high-value care from the American College of Physicians and the Centers for Disease Control and Prevention. Ann Intern Med 2016; 164:425. doi:10.7326/m15-1840
- FDA Drug Safety Communication: FDA advises restricting fluoroquinolone antibiotic use for certain uncomplicated infections; warns about disabling side effects that can occur together. May 12, 2016. Available at: http://bit.ly/3LKT7Bc. Accessed March 30, 2023.
- RJ Zagursky and ME Pichichero. Cross-reactivity in β-lactam allergy. J Allergy Clin Immunol Pract 2018; 6:72. doi:10.1016/j.jaip.2017.08.027
- R Kaur et al. Dynamic changes in otopathogens colonizing the nasopharynx and causing acute otitis media in children after 13-valent (PCV-13) pneumococcal conjugate vaccination during 2015-2019. Eur J Clin Microbiol Infect Dis 2022; 41:37. doi:10.1007/s10096-021-04324-0
- ER Wald and GP DeMuri. Antibiotic recommendations for acute otitis media and acute bacterial sinusitis: conundrum no more. Pediatr Infect Dis J 2018; 37:1255. doi:10.1097/inf.0000000000002009
- AS Lieberthal et al. Clinical practice guideline: the diagnosis and management of acute otitis media. Pediatrics 2013; 131:e964. doi:10.1542/peds.2012-3488
- ME Pichichero. Considering an otitis media antibiotic change. J Pediatr 2020; 222:253. doi:10.1016/j.jpeds.2020.04.021
- Global Initiative for Chronic Obstructive Lung Disease. 2023 GOLD report. Global strategy for prevention, diagnosis and management of COPD: 2023 report. Available at: https://bit.ly/42KEJyV. Accessed March 30, 2023.
- Adult immunization. Med Lett Drugs Ther 2022; 64:161.
- JP Metlay et al. Diagnosis and treatment of adults with community-acquired pneumonia. An official clinical practice guideline of the American Thoracic Society and Infectious Diseases Society of America. Am J Respir Crit Care Med 2019; 200:e45. doi:10.1164/rccm.201908-1581st
- RL Woosley et al. QT drugs list. Available at: www.crediblemeds.org. Accessed December 15, 2022.
- JS Bradley et al. The management of community-acquired pneumonia in infants and children older than 3 months of age: clinical practice guidelines by the Pediatric Infectious Diseases Society and the Infectious Diseases Society of America. Clin Infect Dis 2011; 53:e25. doi:10.1093/cid/cir531
- GS Tansarli and E Mylonakis. Systematic review and meta-analysis of the efficacy of short-course antibiotic treatments for community-acquired pneumonia in adults. Antimicrob Agents Chemother 2018; 62:e00635-18. doi:10.1128/aac.00635-18
- Delafloxacin (Baxdela) – a new fluoroquinolone antibiotic. Med Lett Drugs Ther 2018; 60:49.
- Omadacycline (Nuzyra) – a new tetracycline antibiotic. Med Lett Drugs Ther 2019; 61:74.
- Lefamulin (Xenleta) for community-acquired bacterial pneumonia. Med Lett Drugs Ther 2019; 61:145.
- Corticosteroids in community-acquired pneumonia. Med Lett Drugs Ther 2020; 62:7.
- FDA Drug Safety Communication: FDA review finds additional data supports the potential for increased long-term risks with antibiotic clarithromycin (Biaxin) in patients with heart disease. February 22, 2018. Available at: https://bit.ly/3omrXlH. Accessed March 30, 2023.
- In brief: More fluoroquinolone warnings. Med Lett Drugs Ther 2018; 60:136.